The documents that dictate how a drug is used is called the “label” or the “summary of product characteristics” in Europe or UK. Yesterday, there was a post about biosimilar natalizumab (Tyruko) and reference natailizumab (Tysabri). In the highlights of the paper ( Gelissen et al. Mult Scler Relat Disord. 2026;109:107147. It said
•Discrepant JCV results with the new assay may complicate biosimilar use.
The manufacturers of the drug have to make an assay for the presence of JC virus, which is a risk factor for PML (CNS-brain disease caused by immunosuppression). The originator spent alot of time working out a risk mitigation scheme based on their assay. When the biosimilar assay was created it was not the same and detected more JC virus positivitiy but as they had copied large parts of the Tyrsabri label into the Tyruko lable it means that neuros are advised to do things based on a different assay. Such as do not give natalizumab who are postive for the JC virus. If you have the Sandoz assay you are more likely to be positive than the Biogen assay. So if the cut off was positivity in the assay, then you are less likely to get offered the Sandoz natalizumab.
The manufactures provide this service for free but Biogen are not going to pay or offer their assay to people taking the Sandoz product and likewise Sandoz won’t be doing the same to the Biogen product. This could be a problem for the biosimilar product. But there is another problem which has not been spotted yet but I would add.
Discrepant ADA results with the new assay may complicate biosimilar use.
ADA are anti-drug antibodies (ADA) they develop in people who take natalizumab and in the label there are instuctions to neuro’s what to do. It would not be a problem if the labels/Smpc were different but when they are copied it could be problematic.
Why do I think the biosimilar copies alot, well I did a comparision and if they were students they would be in trouble for plagurism.
So in the label for reference agent it says “The mean ± SD half-life is 11 ± 4 days for the biosimilar the label says exactly the same thing. Which is unremarkable because most of the document is copied. In the UK Smpc it says for the reference produce it is “estimated median half-life was 28.2 days” for the biosimilar it says “estimated median half-life is 28.2 days” for the EU says the same “estimated median half-life is 28.2 days” So the originals are simply copied as the chances of identical results from two separate experiments done years apart are very low and they would be unlikely to be same and to get the same different results in Europe verses USA is weird.
In the EMA assessment report it says the mean half-life ranged between 92.6 (tysabri) and 106 (Tyruko) hours across groups.
It also says “If, after approximately 6 months of therapy, persistent antibodies are suspected, either due to reduced efficacy or due to occurrence of infusion-related events, they may be detected and confirmed with a subsequent test 6 weeks after the first positive test. Given that efficacy may be reduced or the incidence of hypersensitivity or infusion-related reactions may be increased in a patient with persistent antibodies, treatment should be discontinued in patients who develop persistent antibodies“.
So if a person has persientent anti-drug antibodies (i.e. two positive tests six weeks apart), treatment should be discontinued. That is what is says in the summary of product characteristics.
It also says essentially the same in the original in EMA “Disease exacerbations or infusion related events may indicate the development of antibodies against natalizumab. In these cases, the presence of antibodies should be evaluated and if these remain positive in a confirmatory test after at least 6 weeks, treatment should be discontinued, as persistent antibodies are associated with a substantial decrease in efficacy of this medicinal product and an increased incidence of hypersensitivity reactions”
Ok persistent anti-drug antibodies means switch. That could be a problem because the labels say that for reference natalizumab “Approximately 9% of patients receiving TYSABRI developed detectable antibodies at least once during treatment. Approximately 6% of patients had positive antibodies on more than one occasion”
For the biosimilar natalizumab the label says patients in Study MS1 … were tested for antibodies to natalizumab every 12 weeks. The assays used were unable to detect low to moderate levels of antibodies to natalizumab. Approximately 9% of patients receiving natalizumab developed detectable antibodies at least once during treatment. Approximately 6% of patients had positive antibodies on more than one occasion”.
But study MS1 is not with biosimilar natalizumab it is based on the original tysabri study. In the clinical trial of the biosimilar natalizumab the frequency of anti-drug antibody was not 9-10% but a whopping 80% and the number of people with persistent antibodies was not 6% but 55-56%. So if you use the assay developed by the company making biosimilar antibody then an extra 49% will develop persistent antibody and have to switch from natalizumab….So maybe a problem.
It all depends on the assay you are using to determine persistant, all the labels say the same thing the assays developed by the companies and used in the pivotal trials are very different. This i.e., evident when you read this
Chamberlain P, Hemmer B, Höfler J, Wessels H, von Richter O, Hornuss C, Poetzl J, Roth K. Comparative immunogenicity assessment of biosimilar natalizumab to its reference medicine: a matching immunogenicity profile. Front Immunol. 2024;15:1414304.

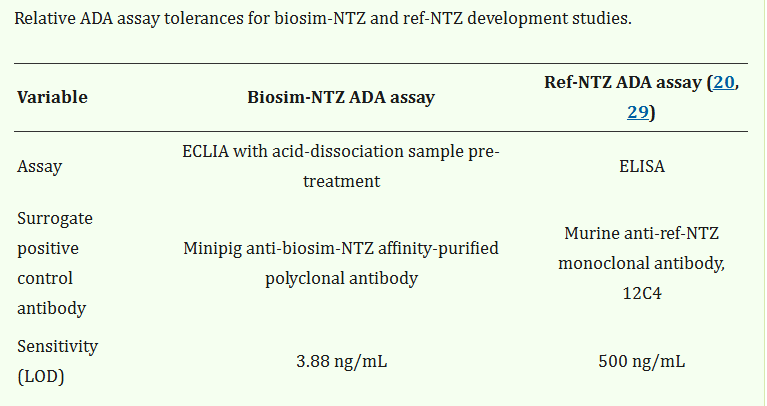
This says to be positive in the reference assay one had to produce over 100 times the amount of antibody detected in the biosimilar assay. The biosimilar assay detected natalizumab/anti-natalizumab complexes as the association between the two were broken using acid conditions.
This means there are many more antibodies detected, it is not the antibody drug that is different but the assay. So both the original and the biosimilar antibody produce persistent antibody in 55-57% of people.

So if you follow the label unless you know that there is a fudge done by the makers of reference natalizumab to under-report the actual number of persistently positive people then you may start to switch people if you use the wrong assay or unless the biosimilar manufacturer has made a new assay. However to do this they will be infringing a patent.
How it works was explained in a paper Calabresi et al. The incidence and significance of anti-natalizumab antibodies: results from AFFIRM and SENTINEL. Neurology. 2007;69:1391-403.
This where the magic 6% comes from…. but the important figure is 500ng/ml is the level reported because this is considered to be a functionally important level. This was defined in a patent
Subramyam M, Amarvadi-L, Wakshull-E, Lynn-F, Panzara M, Barbour RM, Taylor JE. Methods and Products for Evaluating an Immune Response to a Therapeutic Protein. Published in patent WO2006107962 International Filing date : 04.04.2006 So this should expire by 4 April 2026 unless it has been extended.
This says until 2026 the company making the anti-drug assay they could not use 500ng/ml as a reporting level. As antibodies have a half life of about a month, if you can detect antibody at 4ng/ml the chances are a month or two later you are doing to find it. So it all hangs on access to the ADA assay.
The good news is that is not clear where you get reference natalizumab or biosimilar antibodies tested (At BartsMS we have an assay via our NHS service and ProfA has also made an assay)…So you can’t find out if they are presistent or not. I wonder what the regulators would make of this and shows why it is daft for companies to use other company’s data.
The SMPc at the MHRI (UK regulator) needs to be checked and changed if the assay they are providing detects a lot of persistent anti-drug antibodies.
COI: Our work…we wanted to examine this further and contacted both companies…I was not happy.
Disclaimer: These are my views and you can see all this explained once our publication surfaces.
Source: multiple-sclerosis-research.org